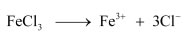Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide. give reason?
![]()
Due to the +I effect of −CH3 group, methylamine is more basic than water. Therefore, in water, methylamine produces OH− ions by accepting H+ ions from water.
![]()
Ferric chloride (FeCl3) dissociates in water to form Fe3+ and Cl− ions.
![]()
Then, OH− ion reacts with Fe3+ ion to form a precipitate of hydrated ferric oxide.