How to draw the resonating structures of H2SO4 and H2SO5 ???
The basic Lewis structure of H2SO4 is

Resonance structure of H2SO4
This structure can be improved by making double bonds with the central sulfur atom. This improves the formal charges. There is more than one possible way to do this. The fact that different arrangements of electrons can be made means that this molecule has resonance.
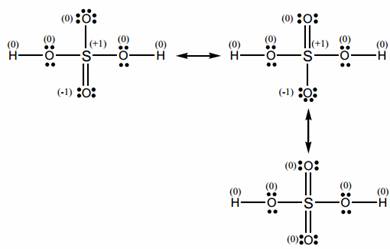
The bottom resonance structure is the best because all the formal charges are zero. We could go on expanding the S octet, but this would start to move the formal charges away from zero; thus we stop here.
Structure of H2SO5
It is known as Peroxymonosulfuric acid, persulfuric acid, peroxysulfuric acid, or Caro's acid. This molecule has (-O-O-) bond (peroxy bond).


